 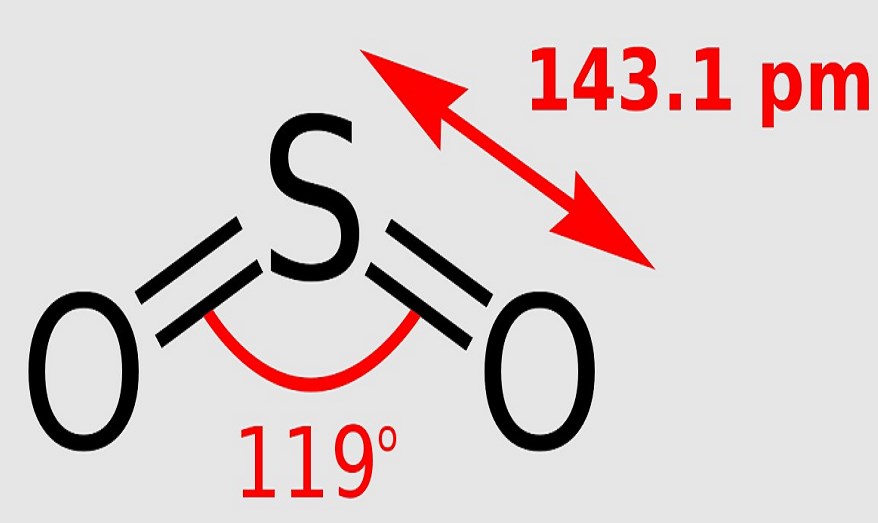
The "smoke" that exits the gas washing bottle is, in fact, a sulfuric acid fog generated in the reaction. The product is sulfur dioxide (SO 2) with some traces of sulfur trioxide (SO 3). A flow-chamber joined to a gas washing bottle (filled with a solution of methyl orange) is being used. An experiment showing burning of sulfur in oxygen. Some sulfur dioxide is also produced by roasting pyrite and other sulfide ores in air. Most sulfur dioxide is produced by the combustion of elemental sulfur. short tons) of sulfur dioxide were used in this way, compared with 150,000 metric tons (165,347 U.S. In the United States in 1979, 23.6 million metric tons (26 million U.S. Sulfur dioxide is primarily produced for sulfuric acid manufacture (see contact process). Īs an ice, it is thought to exist in abundance on the Galilean moons-as subliming ice or frost on the trailing hemisphere of Io, and in the crust and mantle of Europa, Ganymede, and Callisto, possibly also in liquid form and readily reacting with water. The James Webb Space Telescope has observed the presence of sulfur dioxide on the exoplanet WASP-39b, where it is formed through photochemistry in the planet's atmosphere. The atmosphere of Io, a natural satellite of Jupiter, is 90% sulfur dioxide and trace amounts are thought to also exist in the atmosphere of Jupiter. On both Venus and Mars, as on Earth, its primary source is thought to be volcanic. It has been implicated as a key agent in the warming of early Mars, with estimates of concentrations in the lower atmosphere as high as 100 ppm, though it only exists in trace amounts. There, it reacts with water to form clouds of sulfuric acid, and is a key component of the planet's global atmospheric sulfur cycle and contributes to global warming. On other planets, sulfur dioxide can be found in various concentrations, the most significant being the atmosphere of Venus, where it is the third-most abundant atmospheric gas at 150 ppm. Sulfur dioxide is found on Earth and exists in very small concentrations in the atmosphere at about 15 ppb. Occurrence The blue auroral glows of Io's upper atmosphere are caused by volcanic sulfur dioxide. In terms of electron-counting formalism, the sulfur atom has an oxidation state of +4 and a formal charge of +1. There is support for this simple approach that does not invoke d orbital participation. The sulfur–oxygen bond has a bond order of 1.5. Two resonance structures of sulfur dioxide 
SO 2 is a bent molecule with C 2v symmetry point group.Ī valence bond theory approach considering just s and p orbitals would describe the bonding in terms of resonance between two resonance structures. It is released naturally by volcanic activity and is produced as a by-product of copper extraction and the burning of sulfur- bearing fossil fuels. It is a toxic gas responsible for the odor of burnt matches. 
Sulfur dioxide ( IUPAC-recommended spelling) or sulphur dioxide (traditional Commonwealth English) is the chemical compound with the formula S OĢ. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
